Carbodiimide: Difference between revisions
imported>David E. Volk m (Undo revision 100584277 by David E. Volk (Talk)) |
imported>David E. Volk No edit summary |
||
| Line 21: | Line 21: | ||
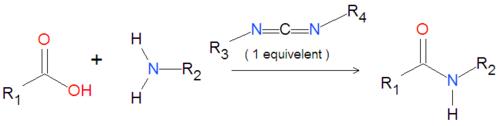
{{Image|Carbodiimide generic reaction.png|left|500px|Coupling of an amide group to a carboxylic acid activated by a carbodiimide.}} | {{Image|Carbodiimide generic reaction.png|left|500px|Coupling of an amide group to a carboxylic acid activated by a carbodiimide.}} | ||
A variety of carbodiimides are commonly used, including EDAC, DIC and DCC, illustrated below. EDAC is particularly useful in aqueous reactions and is sold in a variety of biochemical reagent kits designed for coupling proteins to amines, including amines on the surface of [[quantum dot]]s or [[nanocrystal]]. | A variety of carbodiimides are commonly used, including EDAC, DIC and DCC, illustrated below. EDAC is particularly useful in aqueous reactions and is sold in a variety of biochemical reagent kits designed for coupling proteins to amines, including amines on the surface of [[quantum dot]]s or [[nanocrystal]]. For solid-phase reactions, N,N'-dicyclohexylcarbodiimide (DCC) and N,N'-diisopropylcarbodiimide (DIC) are often chosen as the activating reagent. | ||
== EDAC == | == EDAC == | ||
{{Image|EDAC.png|left| | {{Image|EDAC.png|left|150px|EDAC chemical structure.}} | ||
The reagent [[1-ethyl-3-(3-dimethylaminopropyl)-carbodiimide]], abbreviated as EDAC, EDC or EDCI, is a widely used reagent for coupling water soluble carboxylic acids with water soluble primary amides under aqueous conditions and within a [[pH]] range of 4-6. | The reagent [[1-ethyl-3-(3-dimethylaminopropyl)-carbodiimide]], abbreviated as EDAC, EDC or EDCI, is a widely used reagent for coupling water soluble carboxylic acids with water soluble primary amides under aqueous conditions and within a [[pH]] range of 4-6. | ||
| Line 32: | Line 32: | ||
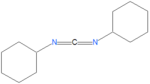
{{Image|Dicyclohexylcarbodiimide.png|left| | {{Image|Dicyclohexylcarbodiimide.png|left|150px|N,N'-dicyclohexylcarbodiimide.}} | ||
== DIC == | |||
The reagent [[N,N'-diisopropylcarbodiimide]], also called 1,3-diisopropylcarbodiimide and abbreviated as DIC, is carbodiimide reagent often used in solid-phase synthesis preferentially over DCC because it is a liquid, and is therefore easier to use then the waxy reagent DCC. DIC is much easier to remove, by extraction techniques, compared to DCC which is otherwise very similar in reactivity. | |||
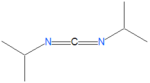
{{Image|DIC.png|left|150px|DCC: Diisopropylcarbodiimide.}} | |||
Revision as of 10:23, 3 October 2009
|
| |||||||
| carbodiimide | |||||||
| |||||||
| Uses: | dehydration reagent | ||||||
| Properties: | activates carboxylates | ||||||
| Hazards: | |||||||
| |||||||
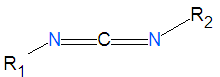
Carbodiimides are a type of dehydrating chemical most often used to activate carboxylic acids for subsequent coupling with primary amines, producing an amide compound. the carboxyl group is often converted to an activated compound by forming an N-hydroxysuccinamide ester or other esters.
A variety of carbodiimides are commonly used, including EDAC, DIC and DCC, illustrated below. EDAC is particularly useful in aqueous reactions and is sold in a variety of biochemical reagent kits designed for coupling proteins to amines, including amines on the surface of quantum dots or nanocrystal. For solid-phase reactions, N,N'-dicyclohexylcarbodiimide (DCC) and N,N'-diisopropylcarbodiimide (DIC) are often chosen as the activating reagent.
EDAC
The reagent 1-ethyl-3-(3-dimethylaminopropyl)-carbodiimide, abbreviated as EDAC, EDC or EDCI, is a widely used reagent for coupling water soluble carboxylic acids with water soluble primary amides under aqueous conditions and within a pH range of 4-6.
DCC
The reagent N,N'-dicyclohexylcarbodiimide, also called 1,3-dicyclohexylcarbodiimide and abbeviated as DCC, was one of the first carbodiimides developed for amide and ester formation from carboxylic acids. It is useful for solid-phase reactions, and has been particularly useful for peptide synthesis because of its high yields for amide forming reactions and its inexpensive cost. Purification difficulties occur using this reagent, and because of this, EDC is often used preferantially to DCC.
DIC
The reagent N,N'-diisopropylcarbodiimide, also called 1,3-diisopropylcarbodiimide and abbreviated as DIC, is carbodiimide reagent often used in solid-phase synthesis preferentially over DCC because it is a liquid, and is therefore easier to use then the waxy reagent DCC. DIC is much easier to remove, by extraction techniques, compared to DCC which is otherwise very similar in reactivity.