Cinoxacin: Difference between revisions
Jump to navigation
Jump to search

imported>David E. Volk (New page: {{subpages}} right|thumb|200px|{{#ifexist:Template:Cinoxacin structure.jpg/credit|{{Cinoxacin structure.jpg/credit}}<br/>|}}Cinoxacin '''Cinoxacin'' is ...) |
imported>David E. Volk No edit summary |
||
| Line 2: | Line 2: | ||
[[Image:Cinoxacin structure.jpg|right|thumb|200px|{{#ifexist:Template:Cinoxacin structure.jpg/credit|{{Cinoxacin structure.jpg/credit}}<br/>|}}Cinoxacin]] | [[Image:Cinoxacin structure.jpg|right|thumb|200px|{{#ifexist:Template:Cinoxacin structure.jpg/credit|{{Cinoxacin structure.jpg/credit}}<br/>|}}Cinoxacin]] | ||
'''Cinoxacin'' is an [[antibiotic]] drug use to treat [[urinary tract infection]]s caused by many aerobic, gram-negative bacteria. It is active against susceptible strains of [[E. coli]], [[Proteus mirabilis]], [[Proteus vulgaris]], [[Klebsiella pneumoniae]], and [[Enterobacter]] species. | '''Cinoxacin''' ('''Cinobac®''') is an [[antibiotic]] drug use to treat [[urinary tract infection]]s caused by many aerobic, gram-negative bacteria. It is active against susceptible strains of [[E. coli]], [[Proteus mirabilis]], [[Proteus vulgaris]], [[Klebsiella pneumoniae]], and [[Enterobacter]] species. | ||
== Mechanism of action == | == Mechanism of action == | ||
Revision as of 12:12, 9 February 2008
Cinoxacin (Cinobac®) is an antibiotic drug use to treat urinary tract infections caused by many aerobic, gram-negative bacteria. It is active against susceptible strains of E. coli, Proteus mirabilis, Proteus vulgaris, Klebsiella pneumoniae, and Enterobacter species.
Mechanism of action
Cinoxacin binds to DNA and interferes with the synthesis of viral RNA and therefore inhibits the production of viral proteins.
Chemistry
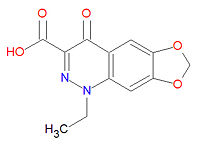
Cinoxacin's IUPAC chemical name is 1-ethyl-4-oxo-[1,3]dioxolo[4,5-g]cinnoline-3-carboxylic acid and its chemical formula is C12H10N2O5 (MW=262.2182 g/mol). It is related to oxolinic acid and nalidixic acid and it is active over the entire urinary pH range.
External links
- Cinoxacin - FDA approved drug information (drug label) from DailyMed (U.S. National Library of Medicine).
- Drug Bank [1]