Ether (chemistry): Difference between revisions
imported>David E. Volk mNo edit summary |
imported>David E. Volk (oxirane ring opening) |
||
| Line 5: | Line 5: | ||
== synthesis of ethers == | == synthesis of ethers == | ||
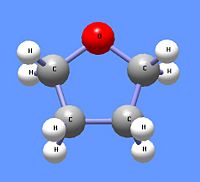
[[Image:Tetrahydrofuran DEVolk.jpg|right|thumb|200px|{{#ifexist:Template:Tetrahydrofuran DEVolk.jpg/credit|{{Tetrahydrofuran DEVolk.jpg/credit}}<br/>|}}Tetrahydrofuran, a cyclic ether.]] | |||
The [[Williamson synthesis]] of ethers uses the [[nucleophilic]] nature of alkoxide ions to react with primary alkyl halides using an S<sub>N</sub>2 [[reaction mechanism]]. Thus, the reaction of sodium isoproproxide reacts with n-butyl iodide to produce the assymetric ether n-butyl isopropyl ether. Primary alkyl halides are used to minimize the E<sub>2</sub> reaction mechanism. The Williamson synthesis can also be used to from cyclic ethers like tetrahydrofuran. To produce [[cyclic ethers]], a primary alkane is used that contains a halide atom at one end and an alcohol on the other end to undergo an [[intramolecular reaction]]. The addtion of the strong base [[sodium hydroxide]] (NaOH) creates an alkoxide ion from the alcohol. The alkoxide end reacts with the halogenated carbon in an S<sub>N</sub>2 mechanism, cyclizing the compound while eliminating the halogen atom. Thus, 4-chloro-1-butanol, in the presence of sodium hydroxide, produces tetrahydrofuran. [[Oxiranes]] are class of cyclic ethers with only two carbon atoms and one oxygen atom in the ring structure. | The [[Williamson synthesis]] of ethers uses the [[nucleophilic]] nature of alkoxide ions to react with primary alkyl halides using an S<sub>N</sub>2 [[reaction mechanism]]. Thus, the reaction of sodium isoproproxide reacts with n-butyl iodide to produce the assymetric ether n-butyl isopropyl ether. Primary alkyl halides are used to minimize the E<sub>2</sub> reaction mechanism. The Williamson synthesis can also be used to from cyclic ethers like tetrahydrofuran. To produce [[cyclic ethers]], a primary alkane is used that contains a halide atom at one end and an alcohol on the other end to undergo an [[intramolecular reaction]]. The addtion of the strong base [[sodium hydroxide]] (NaOH) creates an alkoxide ion from the alcohol. The alkoxide end reacts with the halogenated carbon in an S<sub>N</sub>2 mechanism, cyclizing the compound while eliminating the halogen atom. Thus, 4-chloro-1-butanol, in the presence of sodium hydroxide, produces tetrahydrofuran. [[Oxiranes]] are class of cyclic ethers with only two carbon atoms and one oxygen atom in the ring structure. | ||
== cleavage reactions of ethers == | == cleavage reactions of ethers == | ||
Ethers are readily cleaved with the addition of heat and a strong acid such as [[hydrogen iodide]] (HI). HI is often used because it is acidic enough to protonate the oxygen atom of an ether and the iodide ion is nucleophilic enough to attack the alkyl group of the protonated ether. Thus, [[anisole]] (methyl phenyl ether) can be cleavage to produce phenol and methyl iodide in the presence of HI and heat. | [[Image:Trans-2,3-dimethyloxirane DEVolk.jpg|right|thumb|350px|{{#ifexist:Template:Trans-2,3-dimethyloxirane DEVolk.jpg/credit|{{Trans-2,3-dimethyloxirane DEVolk.jpg/credit}}<br/>|}}One enantiomer of trans-2,3-dimethyloxirane.]] | ||
Ethers are readily cleaved with the addition of heat and a strong acid such as [[hydrogen iodide]] (HI). HI is often used because it is acidic enough to protonate the oxygen atom of an ether and the iodide ion is nucleophilic enough to attack the alkyl group of the protonated ether. Thus, [[anisole]] (methyl phenyl ether) can be cleavage to produce [[phenol]] and [[methyl iodide]] in the presence of HI and heat. Because oxiranes are less stable than other cyclic ethers due to ring strain, [[hydrogen bromide]] (HBr) can be used, without additional heat, to cleave oxiranes such as trans-2,3-dimethyl oxirane. The acid-catalyzed ring-opening of the oxirane enantiomer shown here produces (2''S'',3''R'')-3-bromo-2-butanol. | |||
Revision as of 12:58, 9 January 2008
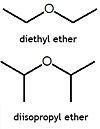
An ether is a chemical compound in which two hydrocarbons are joined together by an intervening oxygen atom. Ethers, particularly diethyl ether, often called simply ether, and tetrahydrofuran (THF) are common solvents for organic chemistry reactions. Diethyl ether was one of the first anaesthetic agents. Many ethers are extremely flammable, and thus use of diethyl ether in the operating room, in the presence of high oxygen levels, in no longer used.
synthesis of ethers
The Williamson synthesis of ethers uses the nucleophilic nature of alkoxide ions to react with primary alkyl halides using an SN2 reaction mechanism. Thus, the reaction of sodium isoproproxide reacts with n-butyl iodide to produce the assymetric ether n-butyl isopropyl ether. Primary alkyl halides are used to minimize the E2 reaction mechanism. The Williamson synthesis can also be used to from cyclic ethers like tetrahydrofuran. To produce cyclic ethers, a primary alkane is used that contains a halide atom at one end and an alcohol on the other end to undergo an intramolecular reaction. The addtion of the strong base sodium hydroxide (NaOH) creates an alkoxide ion from the alcohol. The alkoxide end reacts with the halogenated carbon in an SN2 mechanism, cyclizing the compound while eliminating the halogen atom. Thus, 4-chloro-1-butanol, in the presence of sodium hydroxide, produces tetrahydrofuran. Oxiranes are class of cyclic ethers with only two carbon atoms and one oxygen atom in the ring structure.
cleavage reactions of ethers
Ethers are readily cleaved with the addition of heat and a strong acid such as hydrogen iodide (HI). HI is often used because it is acidic enough to protonate the oxygen atom of an ether and the iodide ion is nucleophilic enough to attack the alkyl group of the protonated ether. Thus, anisole (methyl phenyl ether) can be cleavage to produce phenol and methyl iodide in the presence of HI and heat. Because oxiranes are less stable than other cyclic ethers due to ring strain, hydrogen bromide (HBr) can be used, without additional heat, to cleave oxiranes such as trans-2,3-dimethyl oxirane. The acid-catalyzed ring-opening of the oxirane enantiomer shown here produces (2S,3R)-3-bromo-2-butanol.